Nuvaxovid
Není známo zda se vakcína Nuvaxovid vylučuje. Publicerad idag 0702.
Who Lists 10th Covid 19 Vaccine For Emergency Use Nuvaxovid Strategic Partnership For Health Security And Emergency Preparedness Sph Portal
The Technical Advisory Group for Emergency Use Listing listed Nuvaxovid NVX-CoV2373 vaccine against COVID-19 and Covovax NVX-CoV2373 vaccine against COVID-19.
. Novavaxin Nuvaxovid-koronarokote antaa suojaa SARS-CoV-2 viruksen aiheuttamaa infektiota ja COVID-19 -tautia vastaan. Det proteinbaserade covid-19-vaccinet Nuvaxovid inte ska ges till personer som är 30 och yngre meddelar Folkhälsomyndigheten. The World Health Organization issued an emergency use listing EUL for Nuvaxovid TM following its assessment and approval by the European Medicines Agency EMA earlier.
Age 18 and older. Beslutet är temporärt och gäller från. Nuvaxovid is administered intramuscularly as a course of 2 doses of 05 mL each.
Nuvaxovid contains a version of a protein found on the surface of SARS-CoV-2 the spike protein of the virus that causes COVID-19 which has been produced in the laboratory. Nuvaxovid is composed of purified full-length SARS-CoV-2 recombinant spike S protein that is stabilised in its prefusion conformation. 19 2022 Novavax signed agreements with.
Det eftersom att data från. The addition of the saponin-based. The vaccine contains a version of a protein found on the surface of SARS.
The use of this vaccine should be in accordance with. Rokote ehkäisee erittäin tehokkaasti. 2 Xinhua -- Nuvaxovid the COVID-19 vaccine created by US.
Folkhälsomyndigheten rekommenderar att det proteinbaserade covid-19-vaccinet Nuvaxovid inte ges till personer som är 30 år och yngre. Nuvaxovid has manufacturing sites in the Czech Republic Australia Canada Japan and South Koreas SK. Approved by Health Canada.
On December 20 2021 the. A full list of ingredients for the qualitative and quantitative. Nuvaxovid is indicated for active immunisation to prevent COVID-19 caused by SARS-CoV-2 in individuals 12 years of age and older.
Nuvaxovid is composed of purified full length severe acute respiratory syndrome coronavirus 2 SARS-CoV-2 recombinant spike S protein that is stabilised in its prefusion conformation. Company Novavax should not be given to individuals younger than 30 the Public Health. Nuvaxovid works by preparing the body to defend itself against COVID-19.
How Nuvaxovid works. Cambridge Mass and Osaka Japan April 19 2022 Takeda today announced that it has received manufacturing and marketing approval from the Japan Ministry of Health. Podávání vakcíny Nuvaxovid v těhotenství má být zváženo pouze v případě že možné přínosy převáží možná rizika pro matku a plod.
Unlike mRNA vaccines such as Pfizer and Moderna the Novavax vaccine uses a longer-standing protein-based technology. After the approval of the mRNA vaccines Corminaty BiontechPfizer Spikevax Moderna and the vector-based vaccines Vaxzevria Astra Zeneca and Covid-19 Vaccine Janssen a further. Three large clinical trials showed that Novavax is effective in preventing COVID-19 in people aged 12 years and older.
Nuvaxovid contains a version of a protein found on the. The flu vaccine and the hepatitis B vaccine which. It is recommended to administer the second dose 3 weeks after the first dose see section 51.
Sverige Covid-19-vaccinet Nuvaxovid skulle erbjudas till personer som var tveksamma till vaccinationen. Nuvaxovid is a vaccine for preventing coronavirus disease 2019 COVID-19 in people aged 12 years and older. People who had 2 doses of Novavax were about 80-90 less.
The Nuvaxovid NVX-CoV2373 Novavax vaccine is a recombinant spike S protein nanoparticle vaccine combined with the Matrix-M adjuvant. Novavax Nuvaxovid COVID-19 vaccine. The MHRA can confirm that Nuvaxovid does not contain any components of animal origin.
Nu stoppar Folkhälsomyndigheten användningen bland personer som är 30.

Novavax Covid 19 Vaccine Nuvaxovid Approved By Mhra Gov Uk

News European Commission Grants Conditional Marketing Authorisation For Novavax S Covid 19 Vaccine Nuvaxovid Paul Ehrlich Institut

Novavax S Covid 19 Vaccine Approved For Canadians 18 And Older Cbc News
Ema Doporucila Vakcinu Nuvaxovid Spolecnosti Novavax Zdravezpravy Cz

Informaciya O Vakcine Protiv Covid 19 Nuvaxovid Novavax Australian Government Department Of Health And Aged Care

Novavax Hopes Its Covid Shot Wins Over Fda Vaccine Holdouts The Boston Globe

Chto Iz Sebya Predstavlyaet Novaya Vakcina Ot Koronavirusa Nuvaxovid Raadio 4 Err
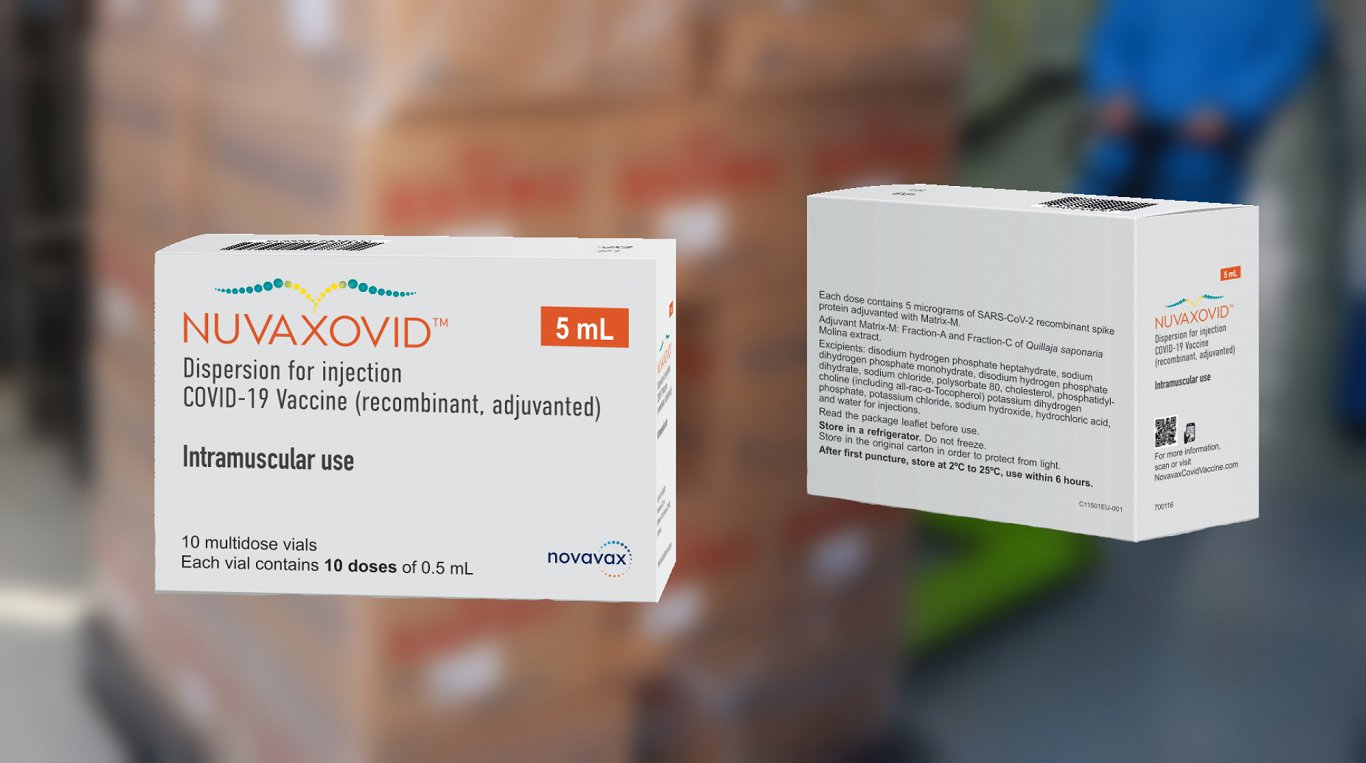
V Estoniyu Pribyli Pervye 18 Tys Doz Novoj Vakciny Ot Covid 19 Nuvaxovid Statya

Novaya Belkovaya Vakcina Nuvaxovid Pribyla V Estoniyu Vaktsineeri Ee

Minzdrav Vakcinoj Novavax Uzhe Mogut Privivatsya Deti S 12 Let Ru Delfi
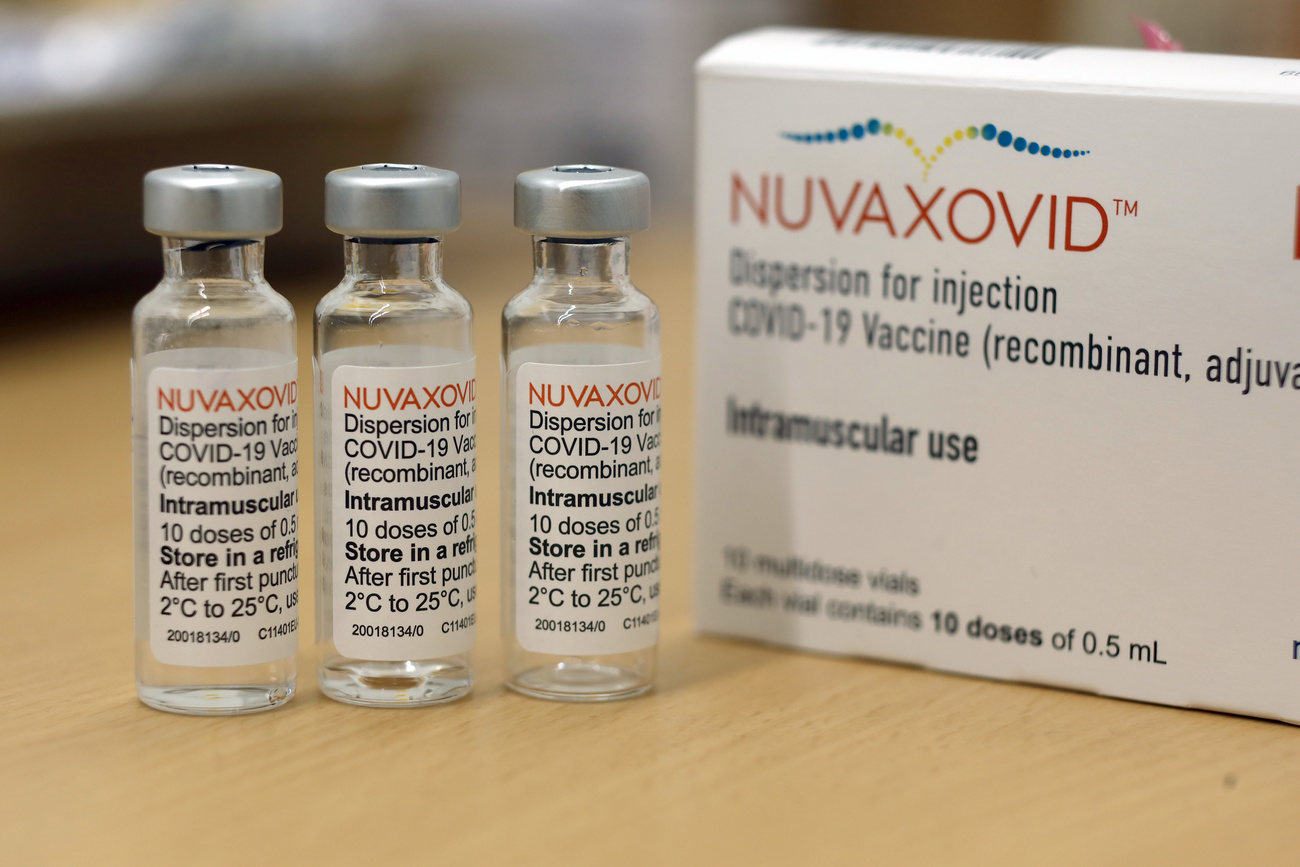
Switzerland Approves Its First Protein Based Covid Vaccine Swi Swissinfo Ch

Sweden Advises Against Covid 19 Vaccine Nuvaxovid For People Under 30 Yrs Investing Com India

Covid 19 Vaccination About The Nuvaxovid Novavax Vaccine Youtube
Novavax Makes One Million Doses Of Nuvaxovid Available For Use In The United Kingdom Pharmtech Focus

What To Know About The New Novavax Vaccine For Covid 19 The Hill
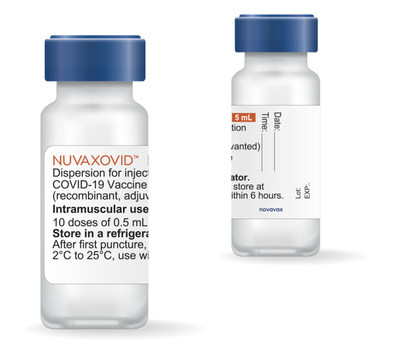
Novavax Announces Shipments Of Its Covid 19 Vaccine To European Union Member States Feb 23 2022
News Conditional Marketing Authorisation Application Submitted For Novavax S Covid 19 Vaccine Nuvaxovid Paul Ehrlich Institut

News Nuvaxovid Novavax Protein Based Covid 19 Vaccine Available On A Limited Basis In Germany Paul Ehrlich Institut

Novavax Covid 19 Vaccine To Be Rolled Out In Australia From Next Month